Anti-TCPb [F39P7F11]
(Cat#: 151868)
Description
| Cat# | 151868 |
| Applications | ELISA, IHC, WB |
| Antigen/Gene or Protein Targets | T-complex protein 1, beta subunit |
| Reactivity | Human, Mouse, Rat, Xenopus laevis and Salmon |
| Relevance | The chaperonins are key molecular complexes, which are essential in the folding of proteins to produce stable and functionally competent protein conformations. One member of the chaperonin group of proteins is TCP1 (chaperonin containing t-complex polypeptide 1, or CCT), but little is known about this protein in tumors. |
| Host | Mouse |
| Immunogen | C-terminal peptide with sequence RKRVPDHHPC conjugated to ovalbumin |
| Subclass | IgG2a kappa |
| Myeloma Used | P3X63Ag8.653 |
| Strain | Balb/c |
| Notes | BLAST analysis revealed 100% identity to the following: Human, Gorilla, Monkey, Marmoset, Mouse, Rat, Dog, Bovine, Hamster, Horse, Pig, Opossum, Platypus, Xenopus, Salmon, Pufferfish, Zebrafish and Stickleback. |
| Research Area | Apoptosis and Programmed Cell Death, Cancer, Cell Cycle |
| References | Characterization and over-expression of chaperonin t-complex proteins in colorectal cancer. Europe PMC ID: 16981251 |
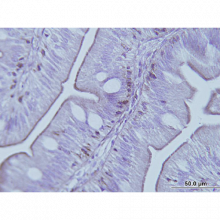
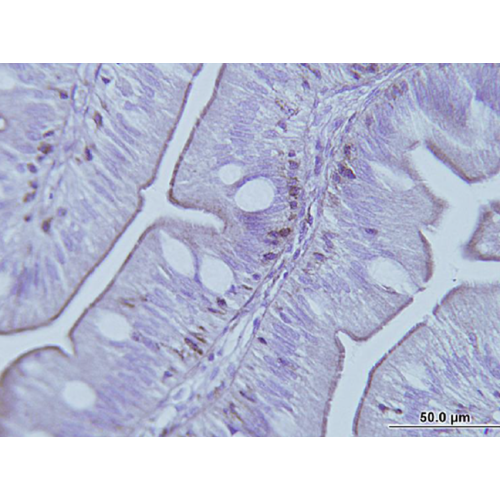
Image: Immunohistochemistry image showing positive staining of Anti-TCPb [F39P7F11] in Gilthead Sea Bream (Sparus aurata). BLAST analysis has shown that peptide immunogen used to develop Anti-TCPb [F39P7F11] is 100% identical in most vertebrates including fish. Immunohistochemistry was performed on the anterior intestine of Gilthead Sea Bream (Sparus aurata). Positive staining was observed in the base of the epithelium (the brush border).
For Research Use Only