Anti-RSV Glycoprotein F [11-2-F3]
| Cat# | 151858 |
| Applications | ELISA, IF, Fn, WB |
| Antigen/Gene or Protein Targets | Respiratory Syncytial Virus glycoprotein F |
| Reactivity | Virus |
| Relevance |
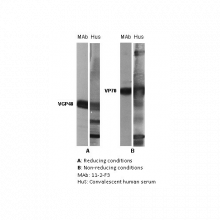
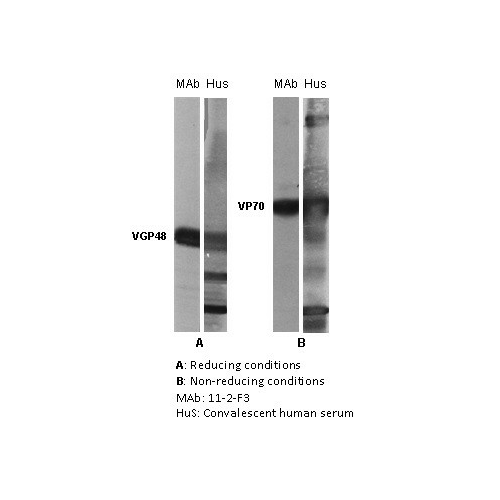
Human Respiratory Syncytial Virus (RSV) is a major cause of lower respiratory tract illness and is the chief cause of hospitalization for respiratory tract illness in young children.The glycoprotein F is located on the surface of viral envelope, its function is to induce fusion of viral envelope with host-cell envelope resulting in syncytium formation. The glycoprotein F (also named VP70, F0 or fusion protein) consists of two components: F1 (also named VPG48) and F2 (also named VGP26) held together by disulphide bonds. The reported molecular weight of the VGP26 component varies between 20 to 26 kDa. Immunoblot using the reduced and unreduced RS virus shows that 11-2-F3 reacts with the non-reduced form of the virus F protein (VP70) and the reduced form (F1). It has a plaque reduction neutralization titer of 10 2.2. |
| Host | Mouse |
| Immunogen | Gradient-purified RSF-44 virus (subgroup A) UV inactivated for 20 minutes at 20oC |
| Positive Control |
Immunoblot: Ag: gradient-purified RS virus (see figure). Indirect immunofluorescence: staining of RSA-2 infected BSC-1 cells |
| Subclass | IgG1 |
| Myeloma Used | P3X63Ag8.653 |
| Recommended Growing Conditions | Dulbecco’s media containing 20% Fetal Bovine serum (DH20) prepared as follows (for final volume of 300ml: 237ml DMEM plus 60 ml Fetal Bovine Serum plus 3ml L-Glutamine). |
| Strain | Balb/c |
| Notes |
This antibody reacts with the RS virus fusion glycoprotein F (also named VP70 or F0 protein), specifically to a subunit of the F protein, F1 (also named VGP48 or 48kDa), from both subgroups A and B of RS virus. This antibody has a high neutralising activity; 90% plaque neutralization titre of 102.2 (Gimenez et al 1996). This property makes it potentially useful for the preparation of reagents for prevention and/or therapeutic use (for example, vaccines or new–generation of man-made antibodies). The characterisation of the epitope of this antibody which induce high neutralising activity will be useful for the preparation of reagents (for example vaccine) to prevent RS virus infection. A RS virus vaccine is not yet available. A vaccine could be used to increase the maternal immunity to RS virus, which would then be passed to the newborn baby to provide passive immunity. The further characterisation of the epitope recognised by this antibody, which possesses a high neutralising activity, will be useful for the preparation of reagents to prevent RS virus infection. New-generation man-made antibodies, based on the 11-2-F3 antibody, could complement the effectiveness of polivizumab (Synagis), which is used to prevent but not treat established RS virus infections (Nature Biotechnology, 2013, 31, 952). |
| Research Area | Virology |
| References |
Immunoblot analysis of the human antibody response to respiratory syncytial virus infection. Europe PMC ID: 3572364 Neutralizing and enhancing activities of human respiratory syncytial virus-specific antibodies. Europe PMC ID: 8705669 Antigenic variation between human respiratory syncytial virus isolates. Europe PMC ID: 3517224 Monoclonal antibodies to human respiratory syncytial virus and their use in comparison of different virus isolates. Europe PMC ID: 6202832 |
Image: The antigen present in all lanes was gradient purified RSN-A2 virus (subgroup A). First antibodies: lanes “mAb”: 11-2-F3 antibody; lanes “Hus”: RS virus convalescent human sera. Panel A: Antigen analysed by electrophoresis using reducing conditions (SDS and mercapthoethanol). Panel B: Antigen analysed by electrophoresis using non reducing conditions (SDS only) (Gimenez et al., 1986). The identity and molecular weight of the protein target of this antibody was validated by including within the immunoblot assay (as a marker) a convalescent serum sample from a RS virus infected patient. The protein specificities of the antibodies induced in the human convalescent serum is described in Gimenez et al. (1987).
For Research Use Only.